SOLVED:Show that the magnetic moment of an electron in a Boht orbit of radius r_{\pi} is proportional to \sqrt{r_{n} .}

IIT JEE - Bohr magneton or magnetic dipole moment of an atom due to revolving electron Offered by Unacademy
![The \"spin-only\" magnetic moment [in units of Bohr magneton, `(mu_B)`] or `Ni^(2+)` in aquenous - YouTube The \"spin-only\" magnetic moment [in units of Bohr magneton, `(mu_B)`] or `Ni^(2+)` in aquenous - YouTube](https://i.ytimg.com/vi/4jAu-kiL0G0/maxresdefault.jpg)
The \"spin-only\" magnetic moment [in units of Bohr magneton, `(mu_B)`] or `Ni^(2+)` in aquenous - YouTube

The value of the magnetic moment of a particular ion is 2.83 Bohr magneton. The ion is - Brainly.in

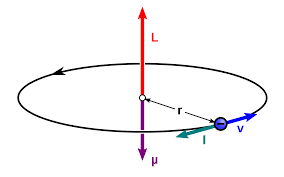
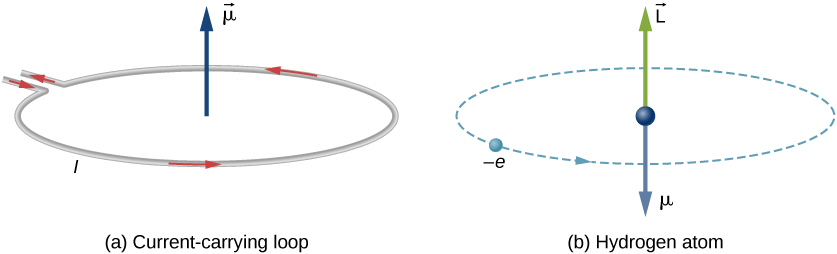
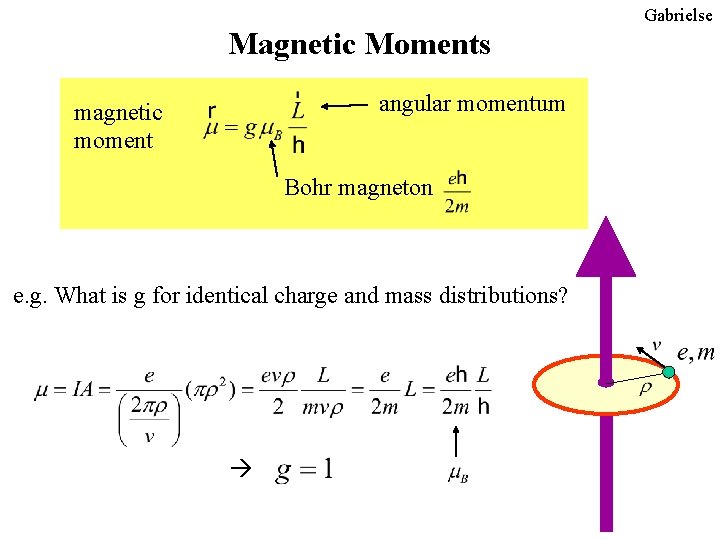
State the relation between orbital magnetic moment of an electron and its angular momentum. Hence find the expression for Bohr Magneton.

The spin-only magnetic moment [in units of Bohr magneton, (mu(B) of Ni^(2+) in aqueous solution would be (atomic number of Ni=28)







![CBSE Class 12 - Bohr Magneton [Unit of Magnetic Dipole Moment] (in Hindi) Offered by Unacademy CBSE Class 12 - Bohr Magneton [Unit of Magnetic Dipole Moment] (in Hindi) Offered by Unacademy](https://edge.uacdn.net/ZWQBR1Q36ZIY2W23CZHL/images/0.jpeg?w=768&fm=webp&q=25)